
FAQ on pharmaceutical pellets
Are you having another FAQ? Just contact us, we can answer.
What material are pellets made of?
Pellet materials for pharmaceutial purpose are:
- Microcrystalline Cellulose
- Tartaric acid (functional)
- Isomalt
- Sugar
- Dibasic Calcium Phosphate
- Polyols
- Lactose-Starch
- Lactose-Cellulose
- Carnauba wax
- Silica
Most pellet materials are seen to be neutral. They do not show mentionable influence on the drug dissolution characteristics. An exception are tartaric acid pellets (TAP). TAPs assist dissolution of weakly basic drugs by serving a localized acidic environment. A good summary is given in an overview article.

Why are sugar pellets often used?
Sugar pellets are long-term available and part of many former formulations for oral dosage forms. However, the trend shows, that chemically inert MCC pellets with lower friability gain importance.
Why do microcrystalline cellulose pellets gain importance?
Microcrystalline cellulose pellets gain importance due to their better properties in drug processes. The chemical inertness and high friability make them becoming perfect starter pellets for multi-particulates.
What is the main purpose of tartaric acid pellets?
The purpose of tartaric acid pellets is to include an additional functionality to pharmaceutical formulations. Tartaric acid pellets serve as starter cores in oral drug formulations (pellet technologies). The chemical properties of tartaric acid serve perfect localized dissolution conditions for weakly base actives.
What are the main functionalities of starter pellets?
Main functionalities of starter pellets are:
- serving as drug carrier in pharmaceutical formulations
- suited for multi-particulate drug solutions
- used for coating and layering with actives and excipients
- allowing time-resolved release profiles
The overall expression for employing pellets in drug formulations is called pellet technologies. Some pellets, such as made of tartaric acid, serve additional functionalities.
main-functionalities-of-starter-pellets
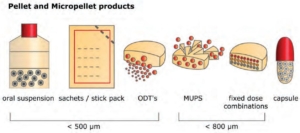
Which products contain pharmaceutical pellets?
Several products contain pharmaceutical pellets. Pharamceutical pellets are found in multipartitular oral dosage forms:
- Oral suspensions
- Sachets / stick pack
- ODT (oral desintegrating tablets)
- MUPS (Multiple Unit Pellet Systems)
- Fixed dose combinations
- Capsules
An excellent overview is listed here.

products contain pharmaceutical pellets
Are pharmaceutical pellets for paediatrics useful?
Yes, definitely pharmaceutical pellets for paediatrics are useful. Advantages are:
- flexible dosing units
- homogeneous drug distribution
- Reduces risks, such as dose dumping
- high customer compliance of adolescents, children, infants, neonates and newborns
The main reason for specialized paediatric medicine are certain differences of the human body interaction with the drug formulation. For example, neonates and infants have increased stomach pH due to decreased acid secretion. Drug uptake and bioavailablity might therefore be different. Modified drug formulations are requested, which do not only consider a change in drug concentration, but also pay respect to dissolution profiles.
Mostly, pharmaceutical pellets for paediatrics are used in pellet technologies.

What is the role of pharmaceutical pellets for geriatrics?
Definitely, pharmaceutical pellets for geriatrics are frequently used in drug formulations. Advantages are:
- Small dosing units
- Formulations, e.g. mini tablets
- Controlled drug release
- high customer compliance for seniors and persons with swallowing difficulties
Additionally, functionalities such as taste masking (which can also serve as a marketing brand tool for product recognition) can be implemented into the drug formulation.
See more information on pellet technologies.

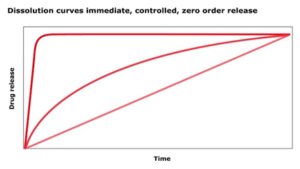
What is an immediate release profile?
An immediate release profile defines an instant drug release, dissolution or uptake. Typical dosage forms are ODTs (oral desintegrating tablets) or sachets / stick packs.
There are several drug release profiles which can be charcterized by dissolution curves:
- immediate
- controlled
- zero order release
In an immediate release profile, the drug is not retarded nor released in a controlled manner. The entire drug dose is released at once (immediately) and available for uptake. The antagonist of immediate release is a retarded or extended release.
A release profile is defiend by the drug formulation. See pellet technologies.
release profiles
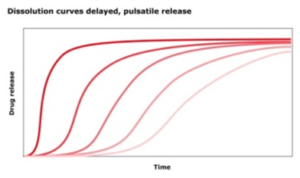
What is a modified release profile?
Other synonyms for a modified release profile are
- extended release profile
- delayed release profile
- Retarded release profile
Relase profiles of drugs allow a controlled dissolution at defined times or locations. The aim is a continuous drug uptake in the body or targeted transport of the drug to the location of highest bioavailability. The other case would be an all-at-once drug release, which is an immediate release.
Find more information about release profiles on pellet technologies. Link to wikipedia.
What are MCC Spheres?
MCC Spheres is pelletized microcrystalline cellulose, are made from certified MCC and pure water. See also: MCC pellets, Cellets.
MCC Spheres are in appearance white or nearly white or beige, hard and almost spherical particles. MCC pellets are odorless, tasteless and are available in sizes from 100 µm to 1400 µm. As excipients, MCC Spheres are versatile used as starter cores in oral fomulations for extended or modified release formulations. They act as neutral starter cores (find here more information about functional starter cores).
In an oral drug formulation, these Spheres are taken for coating and layering of excipients and APIs. This is called pellet technologies. The aims are divers:
- homogeneous drug coating and control
- controlled release
- easy taste-masking of the final drug
- application in multiple unit pellet systems (MUPS)
- new drug formulations for paediatrics
- carrier system for API in jelly formulations
- better customer acceptance and safety
- reduced food effect, variability and dose dumping

MCC Spheres, e.g. Cellets 500.
MUPS – Multiple Unit Pellet System
MUPS is a Multiple Unit Pellet System. They are composed of API layered pellets or starter cores, and matrix fillers.
If pellets are processed into tablets with suitable excipients, MUPS can be achieved. A type of tablet containing MUPS breaks down in the stomach into subunits (in this case individual layered pellets) [1]. It is therefore a special dosage form for solida that combines various properties and characteristics of tablets on the one hand, and capsules filled with pellets on the other hand. This results in several advantages.
First, MUPS are usually smaller than capsules, although they are filled with the same amount of pellets. This increases ease of ingestion and thus has a positive impact on customer’s compliance. MUPS can be divisible, which is impossible with capsules and partly possible with tablets. This allows greater flexibility in therapies. In contrast to coated tablets, drug safety is significantly increased as dose dumping, i.e., too rapid release of a sustained-release dosage form, is minimized. In the case of a tablet, damage to the sustained-release film may be sufficient to trigger dose dumping, whereas a single damaged pellet of an MUPS does not pose a major risk to the patient [3]. Furthermore, inter- and intraindividual variation related to absorption is significantly reduced, as the degree of gastric filling has a lesser impact on drug absorption. Single pellets (< 2 mm) can leave the stomach even with the pylorus closed, whereas monolithic dosage forms (for example, enteric coated tablets) cannot [1].
Find more information about MUPS:
Short information on MUPS – Multiple Unit Pellet System:
- MUPS is an abbreviation for Multiple Unit Pellet Systems
- tablets contain API layered pellets and fillers.
- dose dumping is minimized as in case of pellet-filled capsules.
- high customer patience and easy uptake
- Reduction of inter- and intraindividual variation
Typically, pellets are used as starter beads for API layering. These can be inert pellets, such as MCC pellets (CELLETS®) or functional starter beads, such as tartaric acid pellets (TAP). In some cases, also sugar or wax-like pellets are found in MUPS formulations.

Microscope image of a MUPS tablet, partly opened (3). Copyright: Glatt GmbH.
References:
[1] Opitz, Ulrike. Multipartikuläre Tabletten. Pharmazeutische Wissenschaft. (23) 136-141, 2005 (Link)
[2] Mangesh E. Bhad et al., Int. J. PharmTech Res. 2(1) 2010 (Link)
[3] Glatt GmbH. www.glatt.com. [Date: May 31st, 2013], http://www.glatt.com/cm/de/pharmaceutical-services/formulations-and-technologies/solid-dosage-forms/mups-tablets.html
Are you having another FAQ? Just contact us, we can answer your FAQ.
