Abstract
Several drug substances are known to be extremely bitter. In special for paediatric and geriatric applications, costumer compliance by means of taste acceptance is a highly sensitive topic. Taste masking is therefore an important issue in pharmaceutical industry of oral dosage forms not only to improve the compliance, but it is also used to define a unique taste identification by the costumer (competition advantages). This case study will focus on taste-masking of an API and on the optimization of release in the absorption window of the API.
Modifying the taste
Several methods are available to actively influence, hide or modify taste, such as the initial activation of the active pharmaceutical ingredient (API) by biotransformation [1], the complexation into the cavity of a complexing agent [2] or layering with coating substances [3] and other methods.
Among all these solutions, coating seems to be one of the best understood and most flexible methods. In terms of the API bitterness, mild and extreme bitter APIs are applicable. Additionally, coating [4] is well suited for high dosed API formulations. There are two main approaches: (1) direct coating of the bitter API particles in multiple unit pellet system (MUPS) tablets, or (2) global coating of the tablet which contains the bitter API.
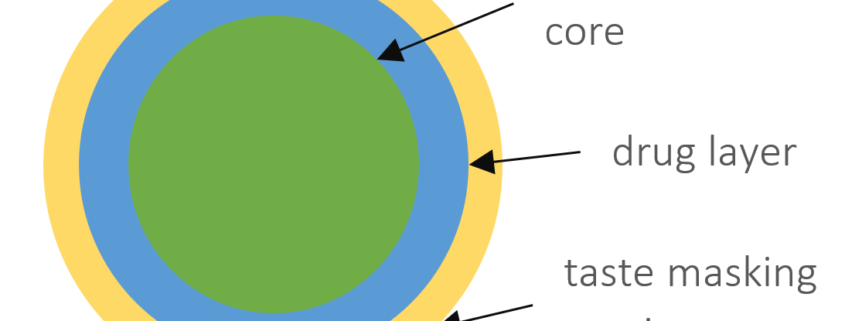
In this case study, we focus on the direct coating of API in a formulation based on starter cores as the unit pellet system. CELLETS® 200 are used as starter cores. They are pellets made of microcrystalline cellulose in size ranges from 100 µm to 1400 µm. In this specific case study, the core size ranges between 100 µm and 200 µm with a defined size distribution. The main advantages of CELLETS® are high sphericity, low friability and optimum in hardness. An API is coated onto the starter cores with a drug load of 5-20 %. The taste-masking coating is performed with Eudragit EPO® so that the resulting particle size remains below 500 µm (Figure 1). Eudragit is the polymer of choice as it ideally prevents API release in oral cavity and allows its release in the absorption window of the API. The final dosage form is a MUPS tablet (Figure 2).
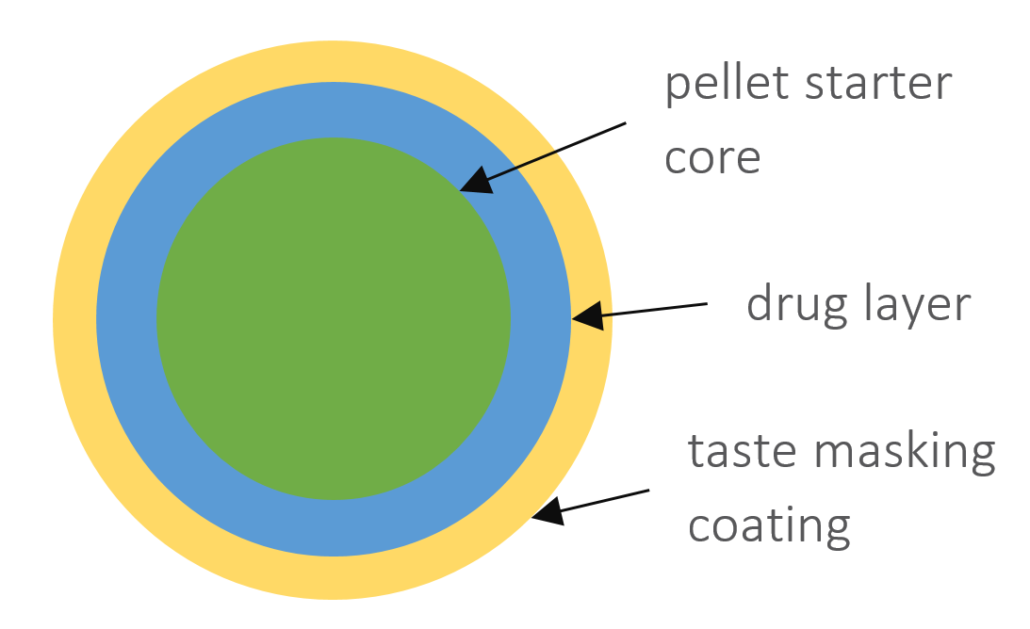
Sketch of a coated pellet starter core (green) with API (blue) and taste masking Eudragit EPO® (yellow) coating. The API drug load is between 5 % to 20 %. The resulting particle size remains below 500 µm.
Compaction is an issue during production, which requires mechanically stable pellets. Coated CELLETS® 200 after compaction still represent as “hard” core pellets. The coated pellets show only little deformation, a reversible compaction behavior and an intact coating.

Sketch of a MUPS tablet. Coated starter cores (yellow) and filling excipient formulation (white).
Here, the MUPS formulation defines a content uniformity with an excellent taste masking at pH 7 (as a measure for the oral cavity) and fast dissolution at pH 1 (representing conditions in the stomach). Figure 3 shows the evaluation of taste masking and dissolution efficiency tests, where the dissolution at pH 7 remains less than 10 % after 10 minutes, and a desired dissolution at pH 1 with a drug release better than 85 % after 30 minutes.
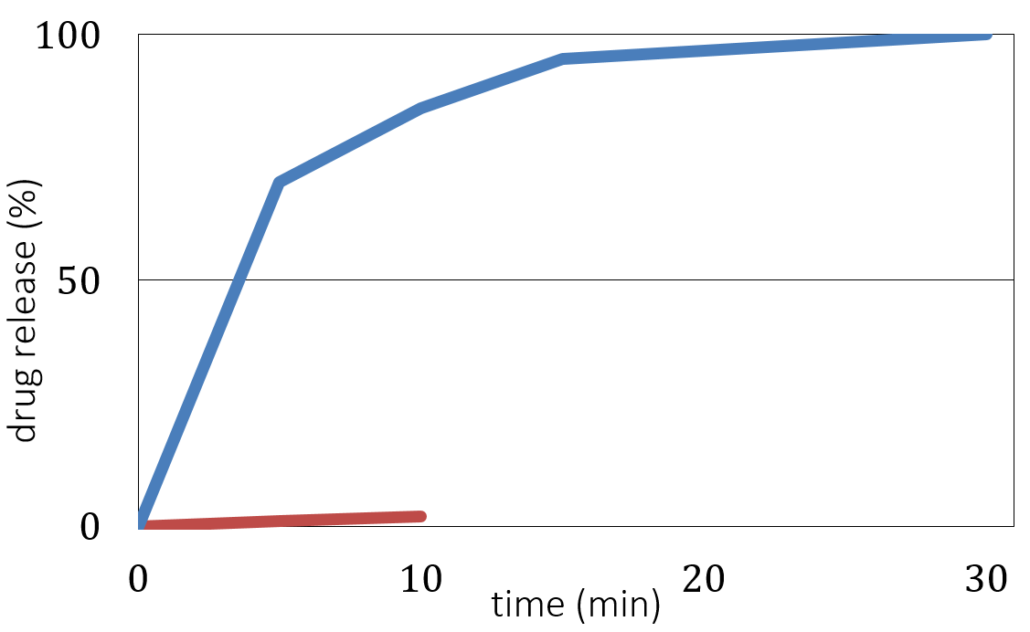
Drug release versus time for coated CELLETS® 200 starter cores. Blue line: in vitro dissolution testing at pH 1 (stomach). Red line: taste masking evaluation at pH 7 (oral cavity).
Summary
Taste masking and modification is crucial for costumer compliance in paediatric and geriatric applications. Several methods are available, such as direct API coating. CELLETS® are perfect starter cores for a successful formulation and simplify taste masking of extreme bitter APIs. CELLETS® properties allow perfect coating abilities due to a high degree of sphericity. CELLETS® of small sizes and narrow size distribution are applicable in MUPS tablets thanks to their low friability and excellent hardness.
References
[4] Hazzah, H.A., EL-Massik, M.A., Abdallah, O.Y. et al. Preparation and characterization of controlled-release doxazosin mesylate pellets using a simple drug layering-aquacoating technique. Journal of Pharmaceutical Investigation 43 (2013) 333–342. https://doi.org/10.1007/s40005-013-0077-0