Characterization of layered pellets forms the basis of advanced pharmaceutical pellet design. These pellets consist of inert cores coated with active drug layers. Their detailed analysis ensures uniformity, strength, and consistent drug release. Layered systems offer precise dosing, extended or modified release, and taste masking. When the solid-state properties of the drug are modified through amorphization, the pellets can show faster dissolution and better bioavailability. In this study, researchers examined how a high-shear granulator can produce pellets that combine amorphized amlodipine besylate and hydrochlorothiazide, focusing on structure, performance, and stability.
Amlodipine besylate normally appears in a crystalline form with high solubility and a melting point near 200 °C. In contrast, hydrochlorothiazide is poorly soluble and melts near 270 °C. Turning them into an amorphous or co-amorphous form breaks down the crystal lattice, improving solubility and dissolution rate. In co-amorphous systems, the two drugs interact through hydrogen bonding, which stabilizes the amorphous state and prevents recrystallization. This interaction increases the dissolution of both drugs and enhances their bioavailability. The study found that partially amorphized drug mixtures in layered pellets improved release rates while maintaining physical stability.
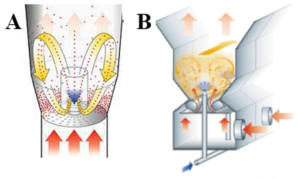
A high-shear granulator creates these layered systems efficiently. Its strong mechanical forces and controlled heat allow uniform coating and induce amorphization at the same time. Because it works without solvents, this process is clean, fast, and suitable for sensitive compounds.
Summary of the Publication
In the study The Development and Characterization of Layered Pellets Containing a Combination of Amorphized Amlodipine Besylate and Hydrochlorothiazide Using a High-Shear Granulator, Mahmoud et al. [1] developed layered pellets by coating microcrystalline cellulose cores (CELLETS®) with drug mixtures in different molar ratios (2:1, 1:1, 1:2). The high-shear granulator (ProCepT 4M8) operated at 1,500 rpm and 60 °C for three hours. The goal was to achieve partial amorphization and study its impact on dissolution and stability. After preparation, the pellets were stored at –20 °C before testing.
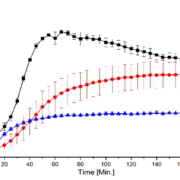
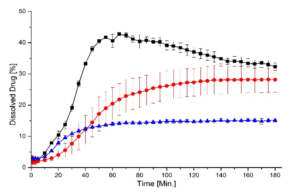
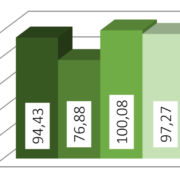
Differential scanning calorimetry showed that amlodipine lost its sharp melting peak, confirming full amorphization. Hydrochlorothiazide retained a broad, weaker peak, meaning it was only partly amorphous. X-ray diffraction supported this: the 2:1 mixture had the lowest crystallinity (26.8 %), while the 1:2 mixture showed the highest (53.6 %). Micro-CT imaging revealed that the drug formed an even layer around the CELLETS® cores. Although some pores appeared, they were inherent to the cores rather than defects from coating.
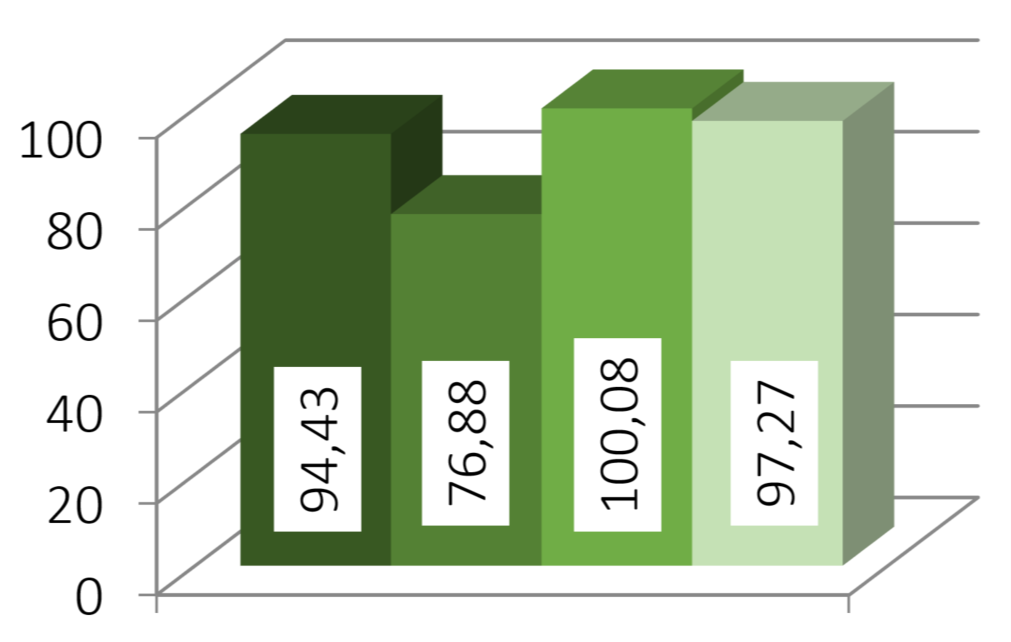
Texture analysis indicated a small increase in hardness—from 19.8 N for plain CELLETS® to around 21 N for layered pellets—showing the coating slightly strengthened the structure. Dissolution testing showed moderate improvement for amlodipine and a strong improvement for hydrochlorothiazide, with release rates increasing up to 2.6 times. The faster release resulted from reduced crystallinity, improved wettability, and closer contact between drug and medium. FTIR spectra revealed broadening and merging of N–H peaks, confirming new hydrogen bonding and lattice disruption. Stability testing over one month showed that 2:1 and 1:1 ratios stayed mostly amorphous, while the 1:2 mixture recrystallized heavily.
Use of CELLETS® in This Study
The authors used CELLETS®, spherical microcrystalline cellulose cores about 1 mm in size, as the foundation for layering. Their smooth and strong surfaces ensured even coating under high shear. Micro-CT confirmed complete drug coverage and consistent thickness. Moreover, the CELLETS® provided the mechanical strength needed to prevent pellet fracture during processing. Their stable core structure helped maintain uniform shape and resistance to deformation.
Conclusion and Outlook
The study proves that solvent-free high-shear granulation can produce layered pellets with amorphized drug mixtures. The characterization of layered pellets showed lower crystallinity, faster release, and stable structure. Using CELLETS® as cores provided excellent mechanical support. The co-amorphous state of amlodipine and hydrochlorothiazide improved dissolution, especially for the poorly soluble hydrochlorothiazide.
Looking ahead, future research should test long-term stability under stress and evaluate in vivo bioavailability. Scaling the high-shear process could make it viable for industrial use. Furthermore, exploring multi-layer systems or combining more drugs could expand the possibilities of characterization of layered pellets in modern pharmaceutical development.
References
[1] Mahmoud et al., Pharmaceuticals 2025, 18(10), 1496; doi:10.3390/ph18101496



 ingredientpharm
ingredientpharm