Abstract
The focus of the current work is to study and demonstrate the impact of the design, the scale, and settings of fluid-bed coating equipment on the differences in pellet coating thickness, which in case of prolonged-release pellets dictates the drug release. In the first set of coating experiments, the pellet cores were coated with the Tartrazine dye with the aim of estimating the coating equipment performance in terms of coating thickness distribution, assessed through color hue. In the second set, drug-layered pellets were film-coated with prolonged-release coating and dissolution profile tests were performed to estimate the thickness and uniformity of the coating thickness among differently sized pellets. In both study parts, film coating was performed at the laboratory and the pilot scale and essentially two types of distribution plate and different height adjustments of the draft tube were compared. The dye coating study proved to be extremely useful, as the results enable process correction and the optimal use of the process equipment in combination with the appropriate process parameters. Preferential film coating of larger drug-containing pellets was confirmed on the laboratory scale, while on the pilot scale, it was possible to achieve preferential coating of smaller pellets using rational alternatives of settings, which is desirable in terms of particle size-independent drug release profile of such prolonged-release dosage forms. […]
Materials
In the first part of the study, neutral MCC pellets (CELLETS 700, IPC Process Center GmbH, Germany) were coated with water solution composed of 8% w/w HPMC 6 mPas (Shin-Etsu Chemical, Japan), 1% w/w Macrogol 6000 (Clariant Produkte GmbH, Glendorf site, Germany), 1% w/w coloring agent Tartrazine (Sigma-Aldrich, USA), and purified water (90%, w/w).
In the second part of coating experiments, API-coated pellets containing Diclofenac sodium were coated with water-based sustained release coating dispersion containing Eudragit RS 30D (9.6% w/w), Eudragit RL 30 D (19.2% w/w) (Evonic Nutrition Care GmbH, Germany), 1.7% w/w triethyl citrate (Vertellus LLC, USA), and 10.4% w/w talc.
Methods
Pellet Film-Coating Experiments with Tartrazin
Coating experiments using Tartrazine dye were performed on two laboratory-sized fluid-bed coaters (GPCG1, Glatt GmbH, Germany and BX FBD10, Brinox d.o.o., Slovenia) and on one pilot-sized (BX FBD30, Brinox d.o.o., Slovenia) fluid-bed coater. In case of both laboratory coaters, the type of distribution plate and the gap between the plate and the draft tube were varied. The pilot-scale setup with three swirl generators and draft tubes was used, while only the size of the gap was varied during coating experiments (Table I). All other process parameters were comparable within each coating process scale. […]
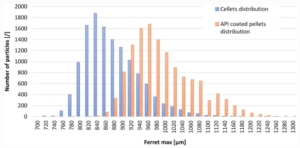
Size distributions of uncoated CELLETS® 700 used in Tartrazine coating experiments and of drug-layered pellets used in PR coating experiments
Conclusion
Considering the results of the coating process evaluation with the dye-coated pellet approach, based on previous research, it can be said that the obtained positive slopes of size preferential coating in the laboratory-scale CW process chamber are within the expected performance of this type of coater design. The values of the slope of the size preferential coating were always lower in the case of the SW distribution plate in comparison with the CW design of the distribution plate. However, within the laboratory-scale coater designs, different performances of swirl generator equipped flat and funnel-shaped distribution plates were identified, the latter exhibiting the least size dependent preferential coating performance. This was attributed to a less expressed dead zone effect enabling mixing and elimination of any segregation in the pellet bed region of the coater. On the pilot film-coating scale, coater equipped with flat SW distribution plates exhibited negative size preferential coating slope, meaning that smaller pellets obtained more coating than larger ones, which is unprecedented result. Moreover, the extent of the negative size preferential coating slope depended on the dynamics of the pressure drop fluctuations. This finding was effectively translated to the prolonged-release coating application, where the right extent of the negative size preferential coating ensures pellet size-independent drug release profiles, thus improving robustness of such multiple unit prolonged-release formulation. By lowering the air flow rate and using bimodal size distribution, rich in smaller drug-layered pellets, led to rather surprising results, where performance of prolonged drug release-coated pellets did not resemble size preferential coating results from the dye coating study part.
These results confirm the fact that we must have a good knowledge of the coater performance characteristics in combination with the process variables and even formulation properties, if we want to produce coated multiple-unit solid pharmaceutical products of the highest quality.