Abstract
Cellets are inert starter cores made of microcrystalline cellulose (MCC). They play an important role in new formulations of solid dosage forms. As a carrier system for actives, the chemical inertness and surface smoothness are crucial parameters. Additionally, high level of robustness and sphericity simplify formulations and technical processes, such as fluidized bed technologies for coating and layering. In a joint study between the University of Hertfordshire and Freeman Technology (a Micromeritics company), the effect of pellets’ size on the behavior in a Wurster process is explained. Wurster fluid bed coating of Cellets with particle size larger than 400 µm is unproblematic. However, decreasing the particle size begins to complicate the coating process. So, powder rheology was used to compare Cellets with different particle sizes in terms of their effect on the powder flow in the Wurster fluid bed coater. For deeper knowledge, we strongly recommend reading investigations by V. Mohylyuk et al. [1]
Materials
Cellets® 90, 100, 200 and 350 (D50-size from 94 µm to 424 μm, Ingredientpharm, Switzerland). MCC powder Avicel® PH-102 (supplied by IMCD UK Ltd., UK) is included in the investigations, as it is widely used in industry and can be used by readers for comparison with other studies.
Wurster fluid-bed
Wurster process is a bottom-spray method, employed as a coating technology, for layering powder-like particles in a fluidized bed system (Figure 1). The process can be separated in different zones of mass flow, such as the down-flow zone or the horizontal transport zone. The flowability in these zones is crucial for homogeneous and efficient coating of the particles.

Figure 1: Wurster process is a bottom-spray method for layering powder-like particles in a fluidized bed system.
Hereby, the size of particles might play an important role. The narrow size distribution of MCC pellets is shown in Figure 2 and Figure 3. Measured data is presented in Table 1.
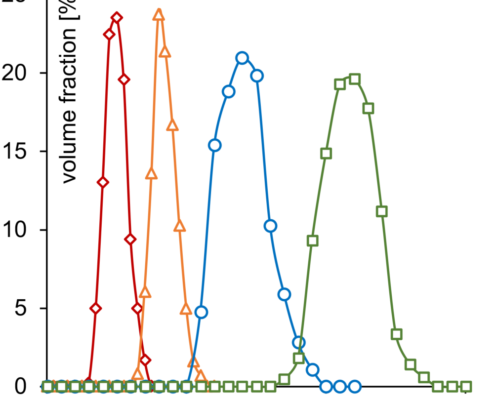
Volume weightened size distribution of Cellets 90 (red, diamonds), Cellets 100 (orange, triangles), Cellets 200 (blue, circles), Cellets 350 (green, squares).
The compact Cellets with fair sphericity, zero friability and a high level of surface smoothness show a fair mass flow rate which is almost independent of particle size at given experimental conditions and was determined by the gravitational funnel method. The reference MCC powder did not flow through the orifice.
| Excipient | PSD [µm] |
Span [µm] |
Flow rate [g/s] |
| Avicel PH-102 | 115a | 1.85 | No flow |
| Cellets 90 | 94b | 0.44 | 1.76 |
| Cellets 100 | 163b | 0.27 | 2.06 |
| Cellets 200 | 270b | 0.34 | 1.89 |
| Cellets 350 | 424b | 0.22 | 1.83 |
Table 1: Particle size distribution (D50) value and span by laser diffraction (a) and digital microscopy (b), and the mass flow rate by gravitational funnel method (5 mm diameter orifice) for the investigated excipients.
Impact of the Cellets’ size
The impact of the Cellets’ size on bulk powder behavior can only be estimated by screening additional parameters. In addition to the mass flow rate, standard pharmacopoeia methods such as bulk/tapped density were initially employed for the characterization of the powder’s properties. This was extended to rotating drum measurements providing the dynamic angle of repose and dynamic cohesivity index. Via powder rheology the conditioned bulk density, basic flowability energy, specific energy, pressure drop, permeability and compressibility (Figure 4) were obtained [1].
By picking the compressibility of Cellets at an applied force of 10 kPa normal stress, two key points need to be mentioned: (a) smaller particle size induces a higher rate of compressibility; (b) Cellets are less compressible than the reference MCC powder.
These findings are part of the open question on powder flow in a Wurster process. It is expected, that Cellets with a lower compressibility will result in better flow behavior in the fluidized bed.

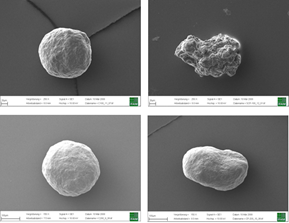
Microscopic imaging of Cellets 100 (top left), 200 (top right), 350 (bottom left) with 1 mm scale bar and 100x magnification. Bottom right: surface of Cellets 350 in 1000x magnification.
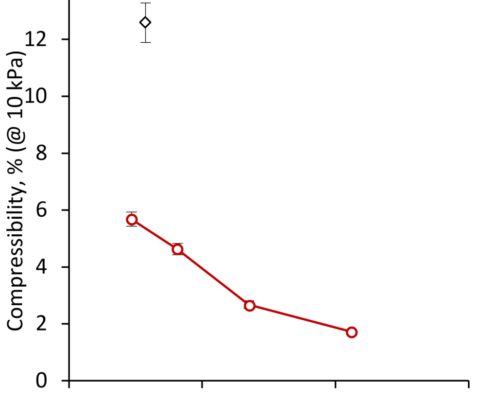
Compressibility at 10 kPa normal stress on Cellets with varying particle size (D50) and Avicel® PH-102.
Summary
The flow of Cellets’ through a Wurster fluid-bed coater is likely to show improved performance as the Cellets’ particle size increases. Among others, a lower compressibility demonstrates a rheological behavior which is superior to MCC.
References
[1] Mohylyuk V, Styliari ID, Novykov D, Pikett R, Dattani R. Assessment of the effect of Cellets’ particle size on the flow in a Wurster fluid-bed coater via powder rheology. J D Deliv Sci Tec. 2019; 54: 101320, doi: 10.1016/j.jddst.2019.101320.

 Ingredientpharm
Ingredientpharm ingredientpharm
ingredientpharm