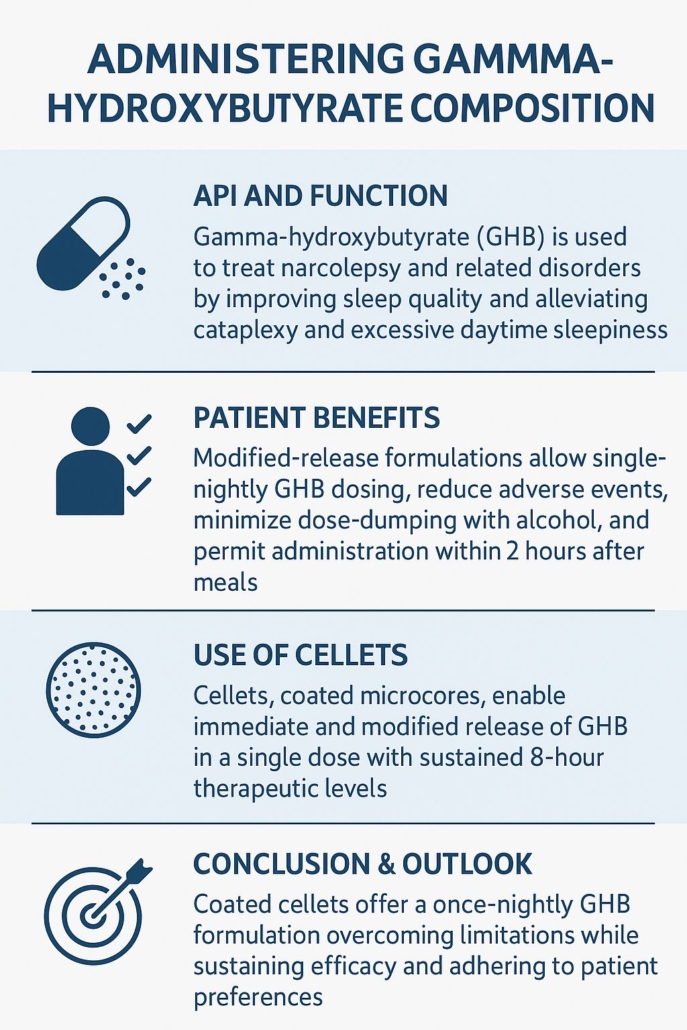
Introduction
Gamma‑hydroxybutyrate (GHB) is an endogenous neurotransmitter also used pharmaceutically—usually as sodium oxybate—for treating narcolepsy and related disorders. It exerts its therapeutic effects by modulating GABA_B receptors and promoting slow-wave sleep, alleviating cataplexy, and reducing excessive daytime sleepiness. Despite its efficacy, current twice-nightly dosing regimens present challenges: dose‑dumping in the presence of alcohol, variable pharmacokinetics depending on food intake, and patient inconvenience. To address these issues, modern formulations—and especially the innovative use of CELLETS® —pursue once-nightly controlled release.
API Benefits and Patient Advantages
Administering gamma‑hydroxybutyrate compositions in a modified‑release format brings multiple patient-centric benefits. A single nightly dose minimizes repeated nighttime awakenings and improves adherence. These formulations exhibit lower peak concentrations (C_max) with sustained therapeutic exposure (AUC)—achieving similar or better efficacy while reducing adverse events such as dizziness or nausea. This consistency is especially meaningful when dosing less than two hours after eating, which often is more convenient for patients; the controlled formulations are more forgiving of fed-state PK variability and less prone to alcohol-induced dose-dumping.
Use of CELLETS® in methods of administering gamma-hydroxybutyrate compositions
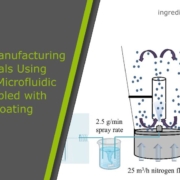
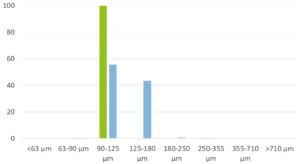
CELLETS® — spherical microcores used in multiparticulate drug delivery—are central to these modern GHB formulations. The patent US 20250186377 A1 introduces coated cellet-based microparticles that incorporate immediate-release (IR) and modified-release (MR) segments within a single unit dose. The MR portion involves CELLETS® (e.g. CELLETS® 90, CELLETS® 100 or CELLETS® 127, and other MCC beads) coated with polymers carrying free carboxyl groups combined with hydrophobic materials (e.g., high melting point waxes), engineered to delay GHB release until intestinal transit. CELLETS® enable precise layering, efficient coating, and reproducible drug release profiles while resisting pH- and alcohol-triggered dose dumping.
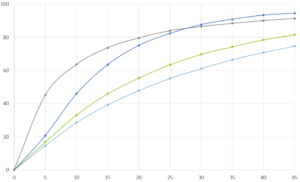
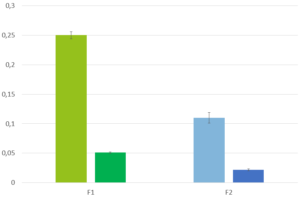
This multiparticulate approach achieves desired PK: IR CELLETS® ensure rapid onset while MR CELLETS® sustain plasma GHB levels up to 8 hours. In contrast to IR liquid sodium oxybate, the coated cellet formulation shows dose‑proportional C_max and AUC across doses of 4.5 g, 7.5 g, and 9 g, with most AEs clustering near C_max but at overall milder intensity. Remarkably, cellet-based formulations maintain comparable therapeutic exposure even with postprandial dosing, offering flexibility not seen in immediate-release forms.
Key Findings
The inventive cellet-based GHB composition delivers both immediate and controlled drug release in one unit, offering dose‑proportional pharmacokinetics and sustained therapeutic levels for 8 hours, under single-nightly dosing. It improves safety by reducing peak‑induced adverse events, lowers risk of alcohol‑related dose-dumping, and allows dosing within two hours after meals. Studies show comparable efficacy to twice-nightly IR sodium oxybate on sleep quality and daytime alertness, with better convenience and adherence.
Conclusion & Outlook
The patented cellet‑based modified-release formulation of GHB marks a significant advancement in administering gamma‑hydroxybutyrate compositions. By incorporating coated CELLETS® that combine IR and MR elements, this approach mitigates common limitations—meal dependency, alcohol interactions, multiple nightly doses—while preserving therapeutic efficacy. For patients with narcolepsy or cataplexy, this translates into improved sleep continuity, reduced daytime symptoms, and enhanced quality of life.
Looking ahead, further clinical evaluation could extend the CELLETS® platform to other formulations of gamma‑hydroxybutyrate salts or co‑therapies (e.g., with sodium valproate), further broadening the therapeutic utility. This modular, multiparticulate delivery system could set a new standard for nightly dosing regimens where controlled pharmacokinetics and patient preferences align.
Patent Details
-
Name/Title: cellet‑based modified‑release gamma‑hydroxybutyrate formulation
-
Patent Number: US 20250186377 A1
-
Year of Patent: 2025
- Patent Holder(s): Not explicitly indicated in the publicly listed data, but associated inventors likely affiliated with pharmaceutical firms focusing on CNS therapeutics (e.g., Jazz Pharmaceuticals or Flamel Ireland).





 Glatt
Glatt